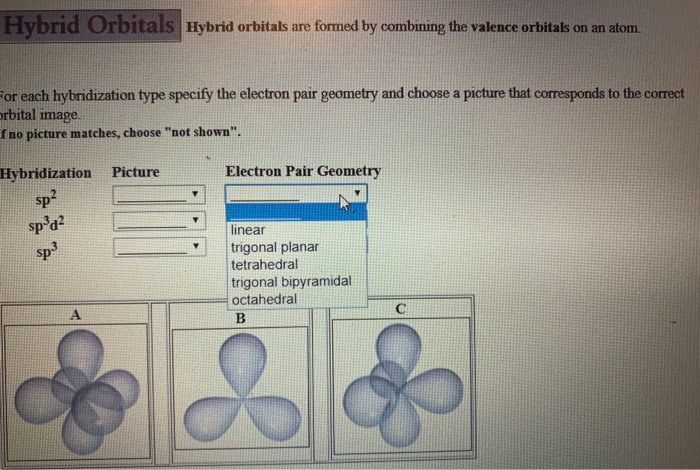
Representing three dimensions on 2D paper (or computer screen) is challenging. The Lewis structure is misleading because it depicts methane as a planar ‘plus-shaped’ molecule, yet we know that methane is a non-planar, tetrahedral molecule. 
Methane (CH 4) depicted by a Lewis structure (left) and dash and wedge notation (right). Depicted on the left in Figure 1 is the Lewis structure for methane (CH 4) showing the central carbon atom singly bonded to four hydrogen atoms: Figure 1. 
The Lewis structure gives us meaningful information about the bonds between atoms, but Lewis structures do not depict how the molecule exists in three-dimensions. | Key Concepts and Summary | Glossary | End of Section Exercises | Lewis Structures with Wedge-Dash Notation | Lewis Structures with Wedge-Dash Notations |
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
